Congratulations to our 2019 5 Minute Presentation Competition Winner Syed Shahabuddin
Congratulations to our 2019 5 Minute Presentation Competition Winner Emily Mitchell
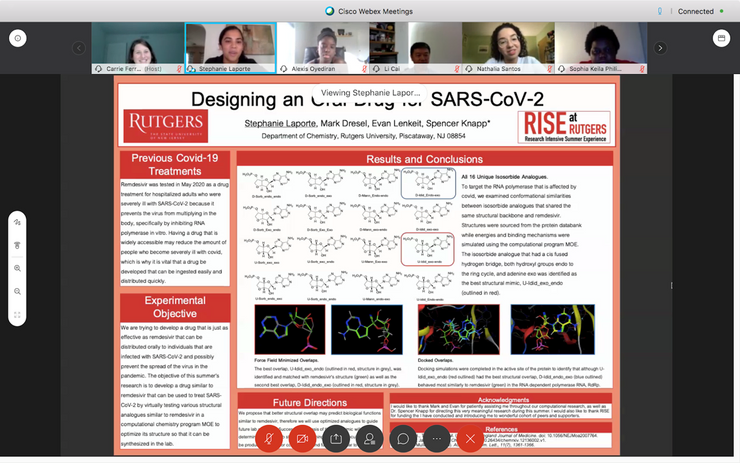
See our 2020 Virtual Symposium presentations. Congratulations to our scholars!
Check out the 2020 Virtual Symposium website where you can find posters and 2020 Scholar-narrated video presentations. Thanks to those who joined us!
Take an Inside Look at Graduate Study at Rutgers
Associate Dean and Chief Diversity Office Evelyn Erenrich gives an inside look at graduate study and paid undergraduate summer research at Rutgers (The State University of New Jersey) on our flagship New Brunswick Campus. Check out the video below.
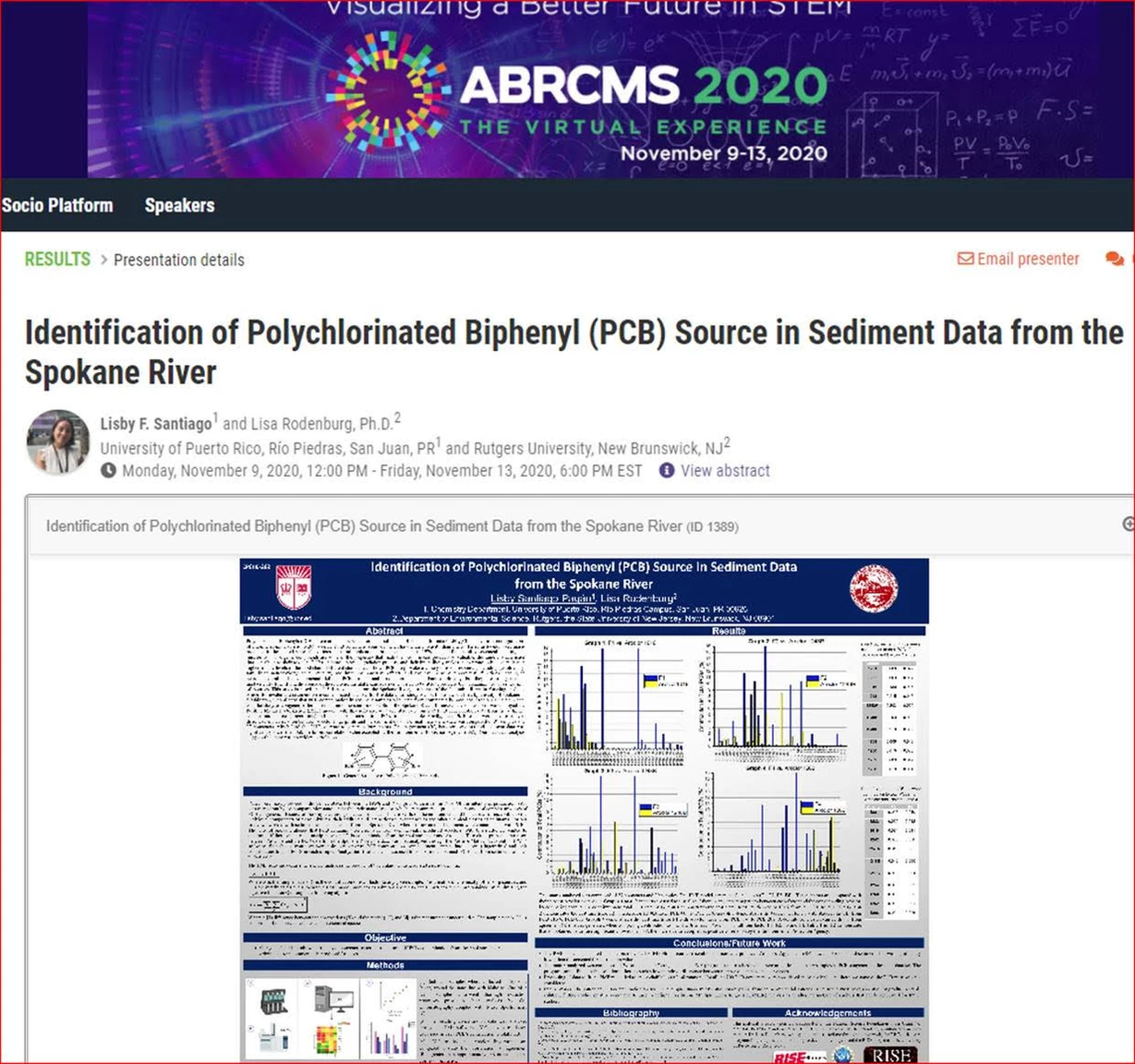
Congratulations to RISE 2020 alum Lisby Santiago for Best Poster Award at #ABRCMS2020!
Register for our Virtual 2021 Summer Symposium
Our Annual RISE Summer Symposium to feature the research of our summer Scholars will once again be virtual. Join us on Thursday, July 29th at 1:30 pm on Zoom using the registration link here.
For information about our keynote speaker and for future updates including the schedule for the day, check out the "2021 Rise Virtual Symposium" page under Events & News.
We hope to see you there!